TAME Trial Media Highlights
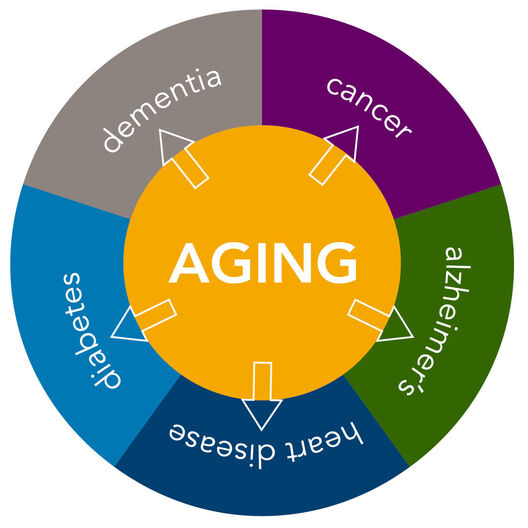
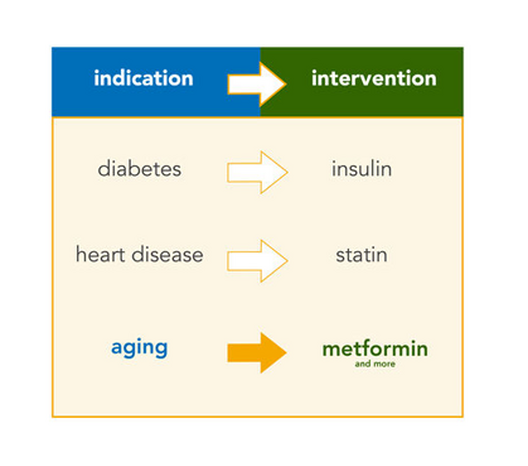
From JAMA to CNN, the Wall Street Journal to Science, the TODAY show to WIRED and The New York Times, the TAME (Targeting Aging with Metformin) Trial has received widespread attention in both scientific journals and popular media worldwide.
Scroll through media highlights at right.